Pain in the Brain
There are several mechanisms that can create a sensation of pain, which has been described as 'an unpleasant sensory and emotional experience in response to perceived or potential tissue damage'. Pain can be the result of peripheral sensitisation from peripheral inflammation, vascular compromise, necrosis, swelling, etc. Importantly, higher centres of the central nervous system not only perceive such sensitization of the peripheral nerve receptors, they can also modulate and control the intensity and tolerability of the perceived sensation through descending modulation at the peripheral receptor and in the spinal cord and through transcortical mechanisms depending on the 'meaning' and 'context given to the pain. Moreover, the higher centres can create a 'state' of perceived 'threat' to the body through emotions such as fear and anxiety. Rather than the brain acting as a filter of unwanted sensation, in the higher centre induced pain state, rumination and magnification of sensations occur to create a pathological state. Paradoxically, representation of body parts such as limbs and individual muscles can reduce in perceived size. In such instances the pain doesn't represent the sensation of pathology but rather pain has become the pathology. Hence, the brain generates pain in the brain, where the pain is perceived to be some sort of non-existent inflammatory or pathological sensation in the periphery. Evidence for this neural plasticity comes from imaging studies, where brain white matter structural properties have been shown to predict transition to chronic pain (Mansour et al 2013, Pain, 154, 10, 2160-2168). Specifically, differential structural connectivity to medial vs lateral prefrontal cortex and connectivity between medial prefrontal cortex and nucleus accumbens has been shown in people with persistent low back pain. In this case the back pain becomes the inciting event and given the persons' structural propensity, establishes specific functional connectivity strength. Some of these changes, in the neural architecture, might be explained by the fact that roughly 5000 of our 20000 genes which make up the human genome are dedicated to chronic pain
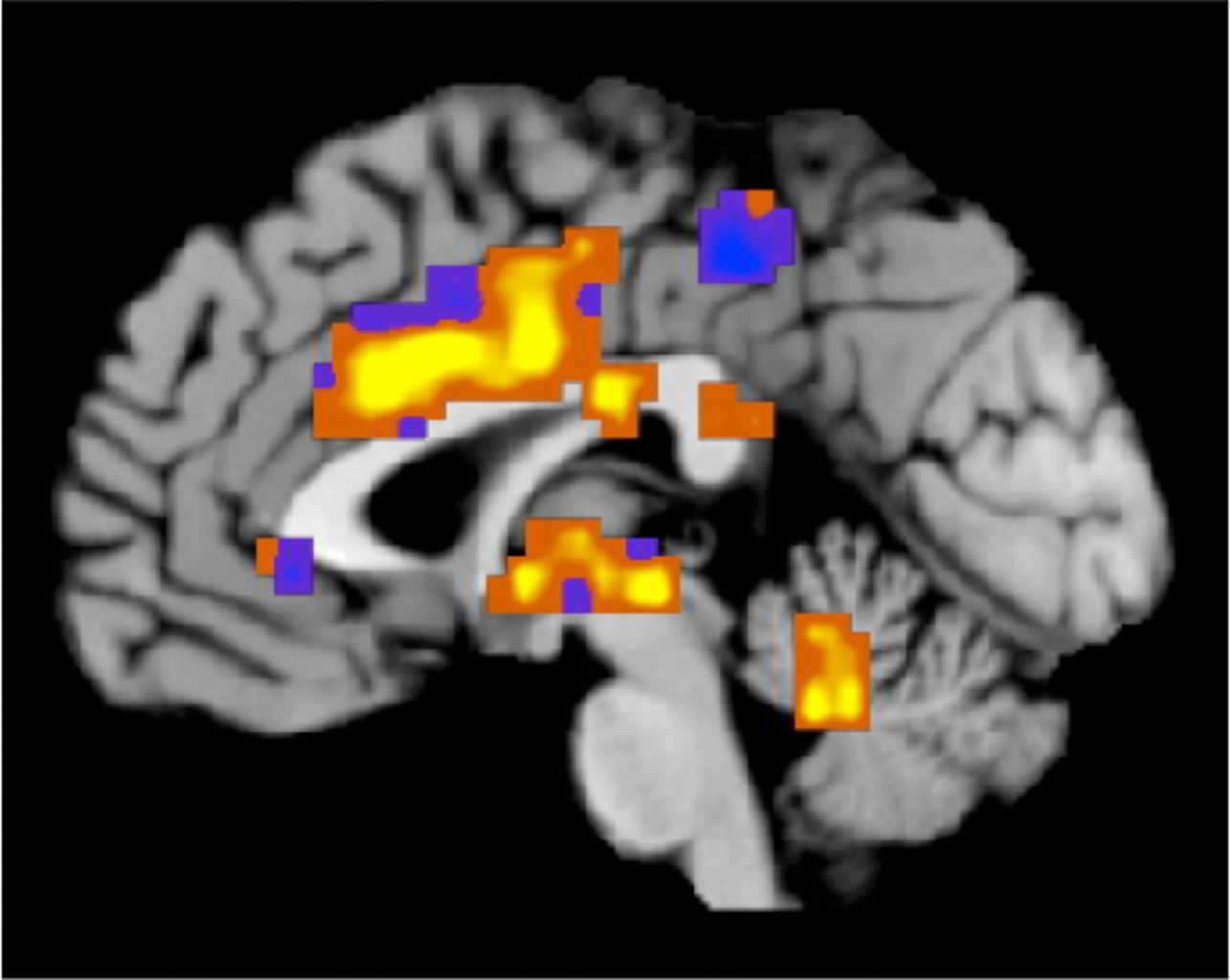
This image created by Tor Wager of the University of Colorado, Boulder shows regions of the "neurologic pain signature," a standard map that can be applied to individual people who may be experiencing pain. The map was developed based on heat pain applied to a group of participants' forearms. Activity in yellow-colored areas is predictive of higher levels of pain, and activity in blue-colored areas is predictive of lower levels of pain. In a provocative new study, scientists reported Wednesday, April 10, 2013 that they were able to “see” pain on brain scans and, for the first time, measure its intensity and tell whether a drug was relieving it. Though the research is in its early stages, it opens the door to a host of possibilities. Scans might be used someday to tell when pain is hurting a baby, someone with dementia or a paralyzed person unable to talk. They might lead to new, less addictive pain medicines. (AP Photo/University of Colorado, Boulder, Tor Wager)
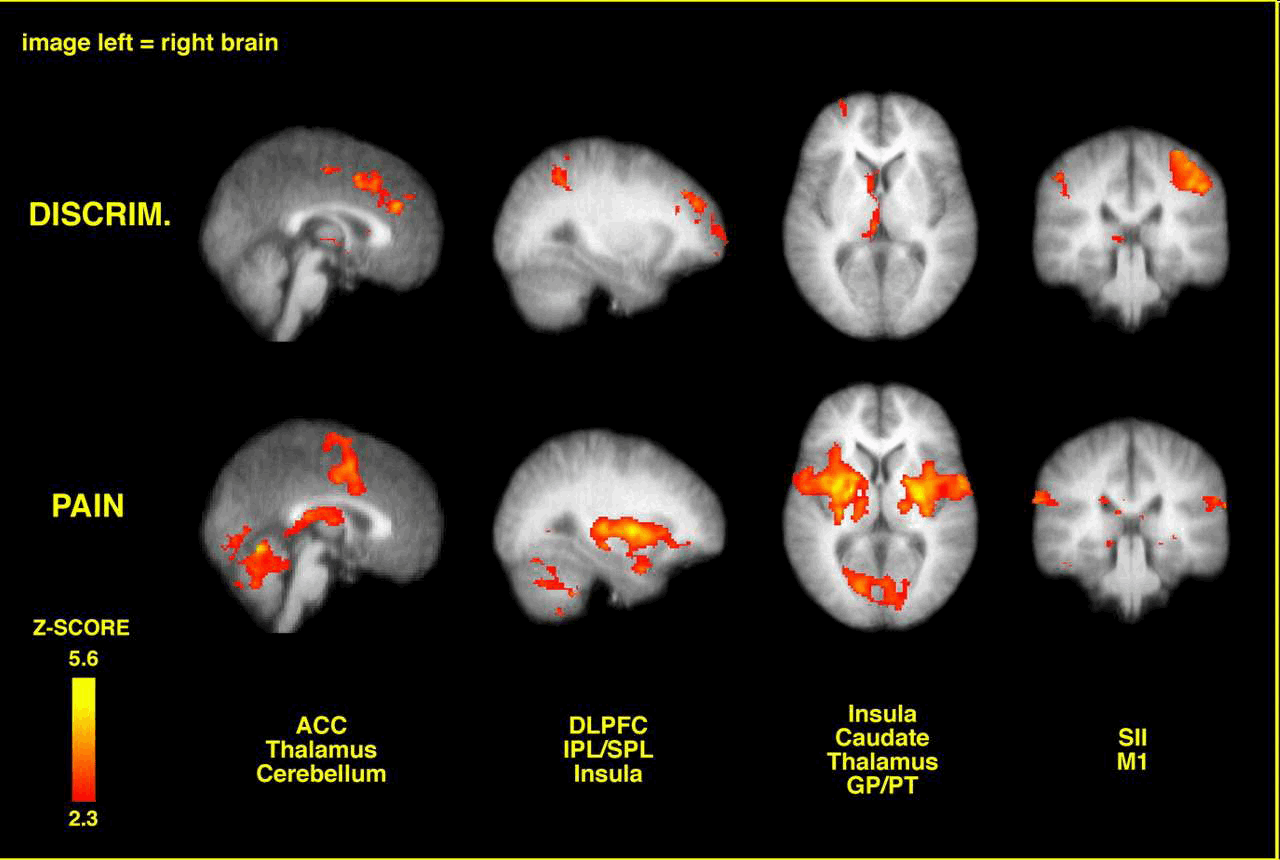
Brain activation related to spatial discrimination of noxious stimuli is distinct from that related to perceived pain. These images are located at x = 0 mm, x = 30 mm, z = 5 mm, and y = –30 mm in standard stereotaxic space. IPL/SPL, Inferior parietal lobule/superior parietal lobule; GP/PT, globus pallidus/putamen; M1, primary motor cortex; DISCRIM., discrimination. (Oshiro et al The Journal of Neuroscience, March 28, 2007, 27(13):3388-3394)
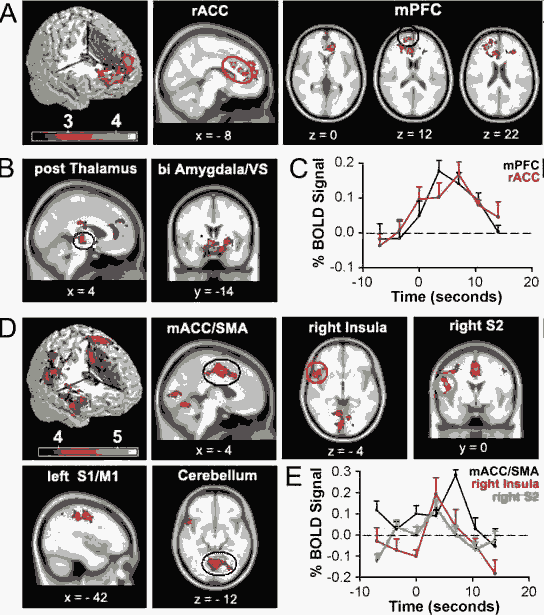
Brain activity for spontaneous CBP shows two distinct patterns: one for phases identified as high pain and another for increasing pain phases. A, Random-effects average brain activity in CBP patients for time periods in which spontaneous pain is high. Activity is limited mainly to the mPFC and rACC. B, Fixed-effects analysis yields additional activations bilaterally in posterior thalamus, amygdala, and ventral striatum (VS). C, BOLD response for peak activations in rACC (10, 22, 28) and mPFC (18, 60, 12) for periods when spontaneous pain is high. Across-subject mean and SEMs are shown. D, Random-effects analysis for periods when spontaneous pain is increasing. Brain activity does not overlap with A and is primarily located in right anterior insula, mACC, and supplementary motor area (SMA), left primary somatosensory (S1), and motor (M1) regions, right secondary somatosensory cortex (S2), and cerebellum. Fixed-effect analysis did not reveal additional brain activity (data not shown). E, BOLD response for peak activations in the mACC (0, 3, 46), right insula (54, 16, –4) and right S2 (46, –20, 28) for time period when spontaneous pain is increasing. Across-subject mean and SEMs are shown. A complete list of activations is found in supplemental tables 2 and 3, available at www.jneurosci.org as supplemental material. Activity maps are presented in MNI space, x, y, and z coordinates in millimeters. Baliki et al, The Journal of Neuroscience, November 22, 2006, 26(47):
Spontaneous chronic Low back pain
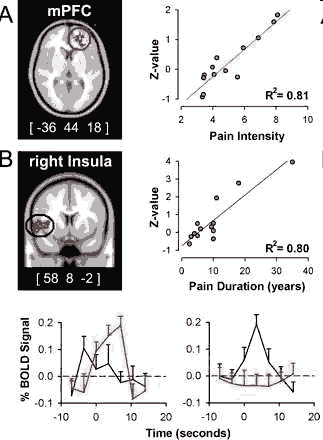
(1) the insular region correlated with pain duration is active during increases in pain whereas mPFC (medial prefrontal cortex) region is not, and (2) the mPFC region best correlated to intensity of pain is active during high pain (for a longer duration than the insula) and in this time period the insular activity is transient and precedes the mPFC activity. This analysis indicates that intensity and duration of CBP can be explained by the differential activity for the two phases of spontaneous pain. (Baliki et al 2006)
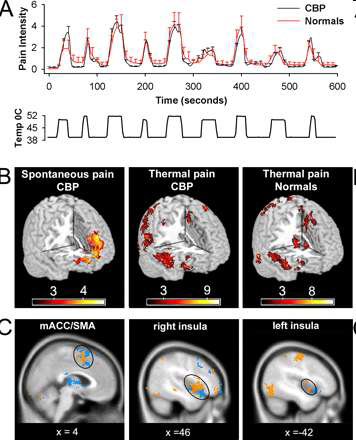
A, Average pain ratings for thermal stimulus applied to the back in CBP patients (n = 11) and matched normal controls (n = 11). There is no significant difference in pain ratings between the two groups. Bottom shows temperature profile of the stimulus applied to the back. B, Random-effects average brain activity for high pain of spontaneous CBP includes mPFC and rostral parts of the anterior cingulate. It is similar to the brain activity pattern observed for spontaneous pain in study 1 (compare Fig. 1A) and does not encompass brain areas activated for noxious heat in CBP (middle) and matched healthy controls (right). C, Similar brain activity patterns are seen for noxious heat applied to the back in CBP (orange) and in healthy normal subjects (blue). It includes the bilateral insular cortex, medial ACC, and supplementary motor areas, in addition to cerebellum and somatosensory regions. Baliki et al The Journal of Neuroscience, November 22, 2006, 26(47)
Nocebo Hyperalgesia
Most people have heard of the power of suggestion activating a positive clinical outcome through the placebo effect. Alternatively, negative clinical outcomes could result in 'learned helplessness' and the 'nocebo effect'
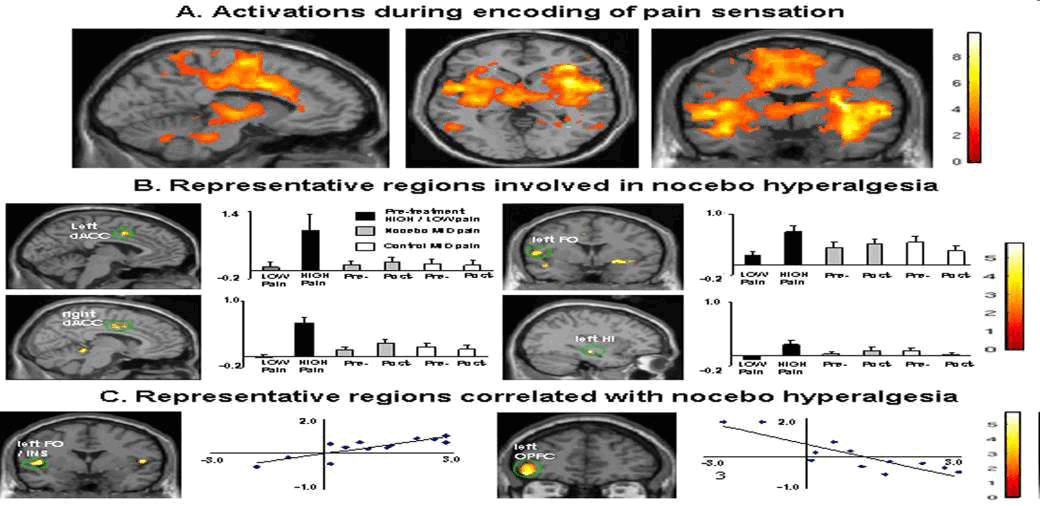
Kong et al ; J Neurosci. 2008 December 3; 28(49)
Brain regions displaying different frequencies of activation between high- and low-sensitivity subgroups
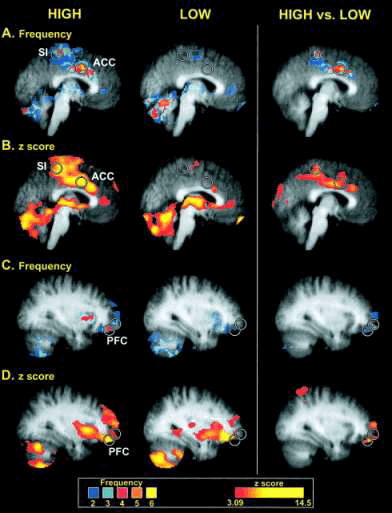
Brain regions displaying different frequencies of activation between high- and low-sensitivity subgroups. Circles are centered on regions where the peak differences between groups were located. Colors in A and C correspond to the number of individuals displaying statistically significant activation at a given voxel (frequency), whereas colors in B and D correspond to the z-score of the subgroup analysis. Slice locations in A and B are - 2 mm from the midline, whereas slice locations in B and C are 32 mm from the midline (in standard stereotaxic space). Structural MRI data (gray) are averaged across all individuals involved in corresponding functional analysis. Coghill R C et al.2003 PNAS ;100:8538-8542
Changes in pain-related activity associated with previous hypnotic training by using suggestions for modulating pain sensation (Upper) or pain unpleasantness (Lower)
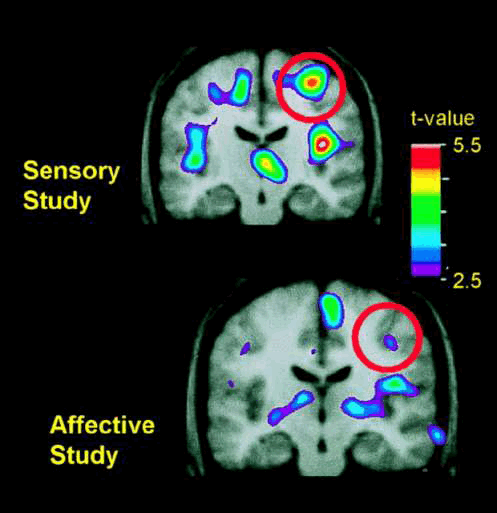
Changes in pain-related activity associated with previous hypnotic training by using suggestions for modulating pain sensation (Upper) or pain unpleasantness (Lower). Both images represent data from control scans, in which no hypnotic suggestions were given. Each image represents the subtraction of PET data recorded when the hand was submerged in thermally neutral water (35°C) from data recorded when the hand was submerged in painfully hot water (47°C). PET data were averaged across 10 experimental sessions in the sensory study (Upper) and, in a different group of subjects, 11 experimental sessions in the affective study (Lower). The PET data are illustrated against the average MRI for that subject group. Bushnell M C et al. PNAS 1999;96:7705-7709
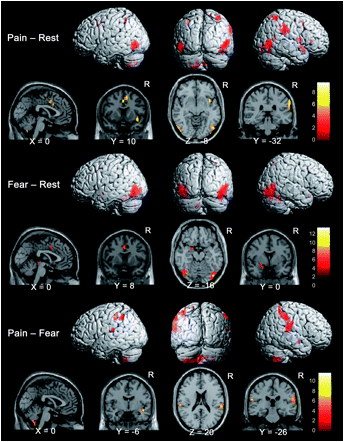
Pain and Fear
Brain activations in each contrast. Activated brain areas in each contrast: pain – rest, fear – rest, and pain – fear conditions. Pain – rest and pain – fear contrasts revealed activations in the SII region and PPC areas and in the affective components of the pain matrix such as the ACC, anterior insula, and cerebellum while viewing images showing painful events. The fear – rest contrast revealed activations in the left amygdala and ACC. Ogini et al (2006) Cerebral Cortex, 17, 5, 1139-1147
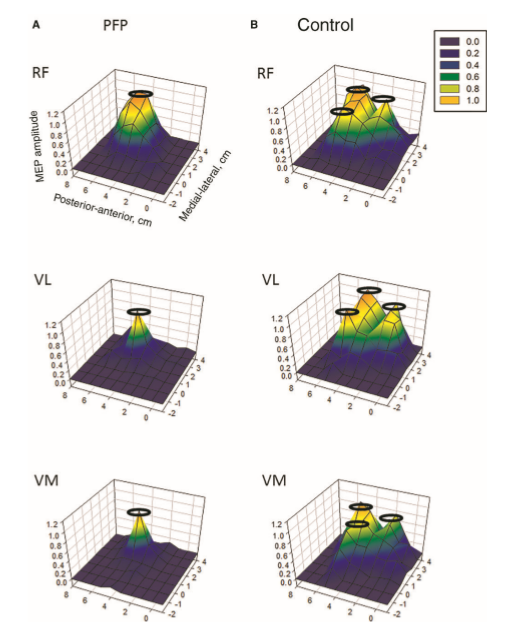
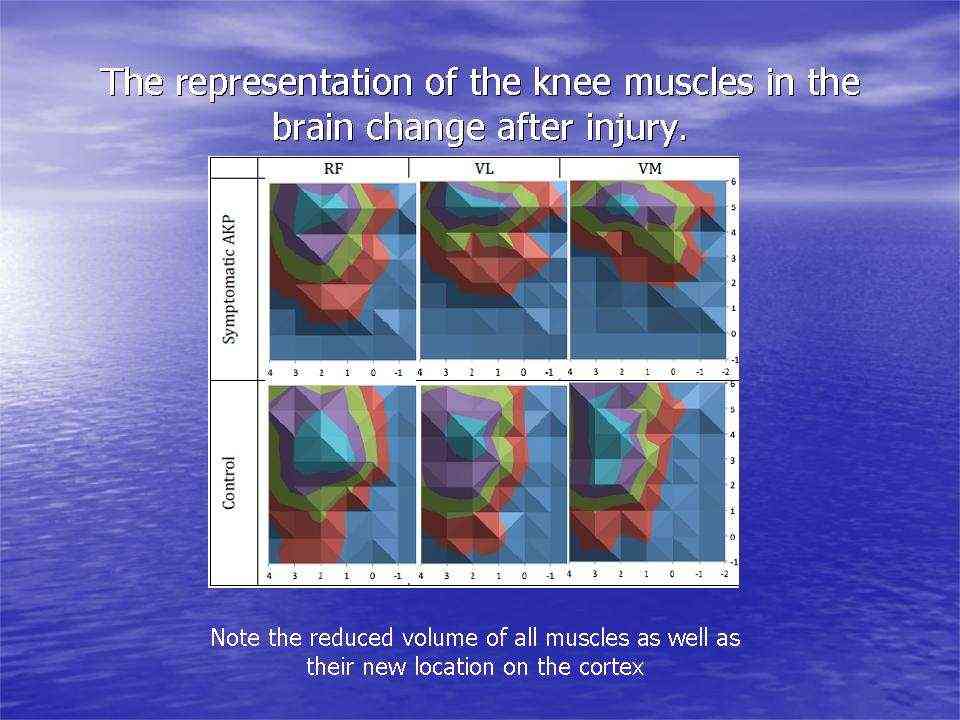
Motor cortical representation and pain
Motor cortical representations have been shown to change in the presence of chronic pain. Trans-cortical stimulation investigations in women with chronic knee pain have shown a reduced volume and location of the quadriceps muscle and especially the VMO (vastus medialis) in the brain. Interestingly, peripheral electrical muscle stimulation can re-establish muscle cortical representation.
Cortical muscle representations in Patellofemoral pain (PFP). Te et al (2017) Pain Medicine, 0, 1-11
The same researcher has shown that maximal tolerable electrical stimulation (eg TENS) of muscles can induce normalisation of the cortical changes through a process called 'smudging'. Transcortical stumilation has also been applied as a cortical 'primer' prior to the application of more traditional therapy such as motor re-training, exercise, and manipulation.
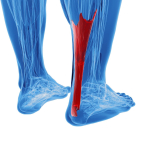
Brain White Matter microstructure of people with Lateral Ankle Sprain
Lateral ankle sprains occur in approximately 70% of the population. These can become chronic and quite disabling leading to declines in physical activity and its concomitant morbidity. Additionally, in the elderly, this can lead to an increased risk of fractures due to falls. Ruptured ligaments have the capacity to alter the somatosensory input to the cortices, thereby altering in turn, the afferent input from the cortices to the periphery, as evidenced by increased use of hip musculature (Doberty et al 2015, Clin Biomech, 30: 129-135), over-reliance of visual input (Song et al 2017, J Sci Med Sport, 20, 10, 910-914) and the frequent use of external stabilisers such as braces and ankle supporting footware. Speculation of pyramidal grey matter changes in the motor cortices have also been made (Terada et al 2016, PM R, 8, 11, 1090-1096). Such altered mechanics has been associated with changes in the central nervous system microstructure of the white matter in the superior cerebellar peduncle (Terada et al 2019, Med Sc Sp Ex, 51, 4, 640-646). Cause and effect cannot be established, however, clinically, emphasis must be placed on both peripheral tissue issues as well as higher centre processes if the chronic cycle is to be broken.
Peripheral Input
Peripheral input is a powerful driver to neuroplasticity. Information gathered by touch, movement and vision, in the context of pain can lead to mal-adaptive plasticity, including the reorganisation of the somatosensory, and motor cortices, altered cortical excitability and central sensitisation. Examples of somatosensory reorganisation come from the work of Abrahao Baptista when investigating chronic anterior knee pain, who not only demonstrated reduced volume of Vastus Medialis but also is cortical translocation to another part of the cortex.
Body illusions are another novel way to promote the normalisation of cortical function through adaptive neuroplasticity. Examples come from people with hand arthritis, whose perception of their hand size is underestimated (Gilpin et al 2015 Rheumatology, 54, 4, 678-682). Using a curved mirror, similar to that in theme parks, the visual input can be increased to perceive the body part as larger (Preston et al 2011 DOI: 10.1093/rheumatology/ker104 · Source:PubMed ) . Irrespective of size, watching a reflection of the hand while performing synchronised movements enhances the embodiment of the reflection of the hand (Whitkopf et al 2017, Exp Brain res, 23, 5, 1933-1944). These visual inputs are thought to affect the altered functional connectivity between areas of the brain thereby affecting the 'pain matrix'.
Another, novel way of looking at movement and pain perception is the concept of the motor engram. This has been defined as motor skill acquisition through the modification and organisation of muscle synergies into effective movement sequences. The learning process is thought to be acquired as a child through experientially based play activity. The specific neural mechanisms involved are unknown, however they are thought to include
- motor map topography reflecting the capacity for skilled movement
- reorganisation of motor maps in a manner that reflects the kinematics of acquired skilled movement
- map plasticity is supported by a reorganisation of cortical microcircuitry involving changes in synaptic efficacy
- motor map integrity and topography are influenced by various neurochemical signals that coordinate changes in cortical circuitry to encode motor experience (Monfils 2005 Neuroscientist, 11, 5, 471-483).
Interestingly, it is an intriguing notion that accessing motor engrams from patterns acquired prior to the pain experience might lead a normalisation of brain activity. My personal experience of severe sciatica with leg pain, sleepless nights and a SLR of less than 30 degrees, happened to coincide with training my 9 year old sons soccer training. I was noticing that the nights after i trained the children, I slept much better and my range of movement improved. I commenced a daily program of soccer ball tricks which i had been showing the kids, including 'juggling', 'rainbows' and 'around the worlds'. Eventually, I even took up playing soccer again after a 30 year absence from the sport. Other than new activity related pain issues (DOMS), four years on, the sciatica hasn't returned. I can only conclude that this activity activated dormant childhood motor engram, worked on global balance, mobilised my nerve, encouraged cross cortical activity and turned my focus into functional improvement.
Alternatively, the altered pain state can result in a hostage like situation, whereby the pain takes control. Similar to the 'Stockholm Syndrome' where the hostage begins to sympathise with their captors, so do some peoples brain states, where it begin to sympathise with the pain, creating an intractable bondage and dysfunctional state.
Conclusion
The research findings present an interesting dimension in the treatment of musculoskeletal disorders. Whilst the Musculoskeletal and Manipulative Physiotherapist use their hands to treat the biomechanics and neurophysiology of the peripheral nervous system and its pathology, the use of exercise regimes and cognitive strategies inducing central nervous system involvement is always inherently important. Since the brain is use to instigating motor responses, it is plausible that the 'motoric intent' determines the significance of sensations from the peripheral nervous system, rather than what was commonly believed to be that sensation determined motor responses. However, in some instances, the brain can magnify the pain through increased arousal towards a 'perceived or real threat', with the consequence that the person only ever sees the 'negative', where 'the bucket is only ever 1/2 empty'. Manifestations of 'learned helplessness' may occur, giving rise to severe deconditioning through 'fear-avoidance' behaviour which affects the immune system. It is important for the person to understand the way pain is processed in the brain and how thought processes can turn a 'bad' pain into a 'good' pain, or alternatively, a normal sensation can be turned into a pathological one due to rumination and magnification of innocuous stimuli.
Hence, higher centres need to be engaged in rational cognitive processing. This may be reflected in something as simple as establishing a dialogue with the client, where 'concerns, fears, beliefs and anxieties' can be ascertained, and whereby explanations can be given, misconceptions are nullified, and empathy can be shown towards the person suffering an affliction. The brain is like a highly tuned orchestra where multiple brain functions are integrated for movement. Client expectations and perceptions will drive the outcome, it is up to the clinician to bring clarity into the management process, by introducing the conductor to the orchestra. Please read the sections about chronic low back pain and the immune system elsewhere on this website.
Updated : 18 July 2021
ini et al (2006) Cerebral Cortex, 17, 5, 1139-1147